Capture the issue
Record nonconformities, complaints, audit findings, observations, and improvement opportunities with consistent, searchable data.
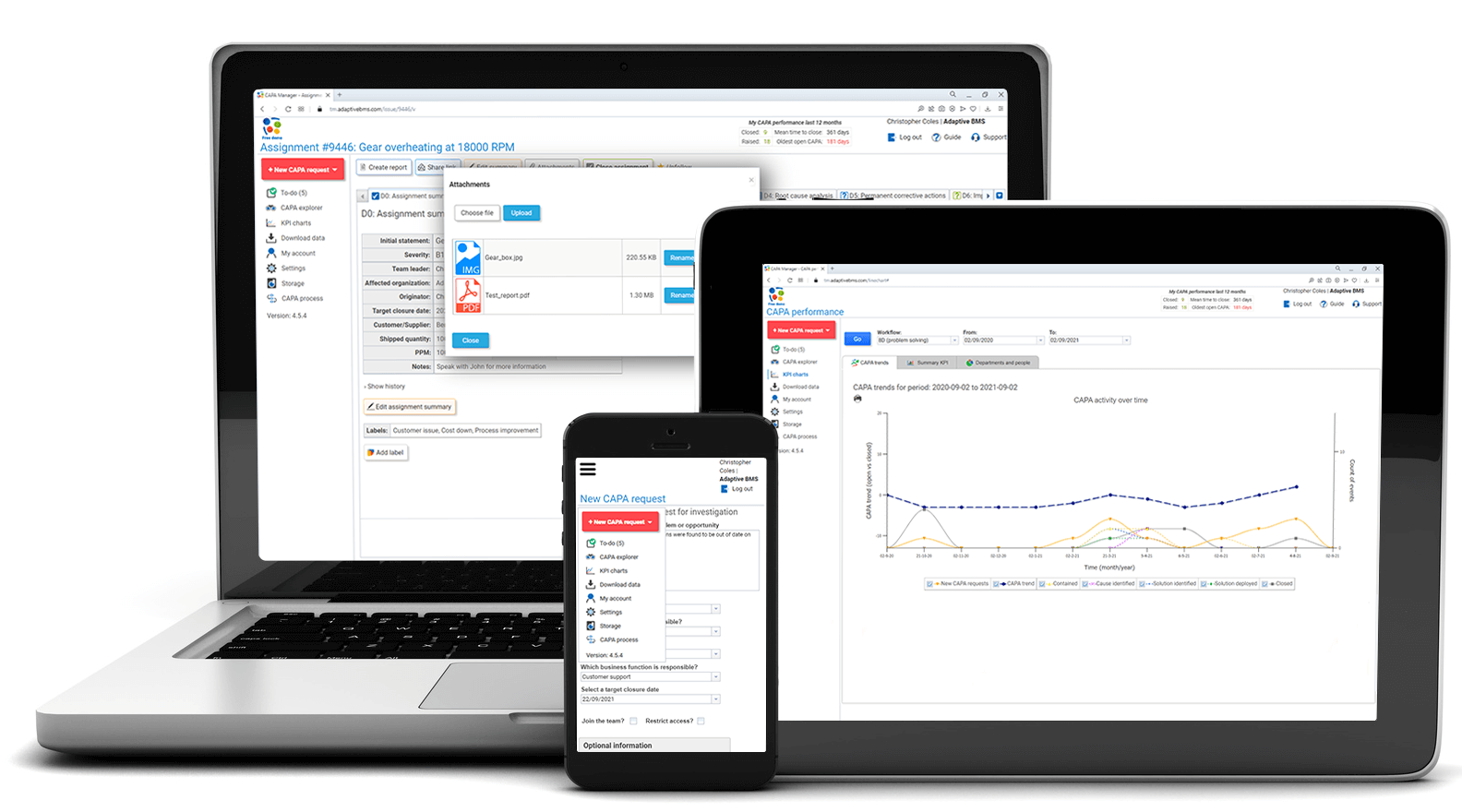
CAPA Manager software
Manage corrective actions, root-cause investigations, approvals, evidence, and closure in one clear cloud-based workflow.
Record nonconformities, complaints, audit findings, observations, and improvement opportunities with consistent, searchable data.
Assign owners, gather evidence, identify root cause, plan actions, and keep every decision connected to the investigation.
Use approvals, verification, digital signatures, and complete history to prove that actions were completed and effective.
CAPA Manager helps teams control corrective and preventive action from first report through investigation, action planning, effectiveness review, and final closure.
Improving quality and maintaining compliance requires an effective Corrective and Preventive Action management system (CAPA). The CAPA process allows teams to capture and record problems like nonconformities, deviations, observations, and improvement opportunities, then assign responsibility for identifying the root cause and implementing corrective actions.
The CAPA process can improve operating processes from manufacturing to marketing. CAPA Manager enables organizations to manage quality performance and improvement across the entire operation quickly and effectively, enhancing quality, safety, and overall process compliance.
Automate the assignment, notification, investigation, reporting, and approval of corrective actions. CAPA software error-proofs the control of the corrective action process from initiation to investigation and closure.
CAPA Manager helps your organization take control of corrective action and improvement processes with a fully compliant, easy-to-use software solution designed to boost team engagement and reduce overhead.
Get started in minutes. CAPA Manager is cloud-based CAPA software with no installation delay, clean screens, and built-in workflows for quick adoption.
Track quality incidents from initial reporting through investigation and corrective action deployment, with a real-time view of tasks in progress.
Use one system for workflow control, notifications, investigation tools, approvals, evidence, reporting, and secure records.
Support CAPA, 8D, RCCA, A3, DMAIC, PDCA, audits, HSE events, and improvement projects.
Keep action owners, reviewers, and approvers moving with timely reminders and escalations.
Use 5 Whys, Ishikawa analysis, risk matrices, prioritisation, and labels to understand recurring issues.
Protect approvals and closure with secure, traceable sign-off steps for validated records.
Attach files, documents, images, and supporting evidence directly to investigations and actions.
Track progress, overdue work, improvement trends, and data needed for audits or management review.

Effective corrective action depends on understanding cause, risk, and recurrence. CAPA Manager gives teams practical RCCA tools so investigations move beyond symptoms and into sustainable prevention.
Get access to CAPA Manager and see how quickly your team can begin managing corrective actions in a structured workflow.
No sales calls. No Credit Card Required. Just instant access to your CAPA Manager account.